All products
Antibodies
p-c-Myc Antibody (Thr 358)

Western blot analysis of phosphorylated c-Myc expression in untreated (A) and UV-treated (B) HT-29 whole cell lysates.
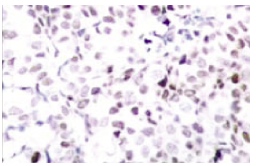
Immunoperoxidase staining of formalin-fixed, paraffin-embedded human breast carcinoma tissue showing nuclear staining.
Product Profile
| Product Name | p-c-Myc Antibody (Thr 358) |
|---|---|
| Antibody Type | Tags Antibodies |
| Modification Notes | c-Myc-, N-Myc- and L-Myc-encoded proteins function in cell proliferation, differentiation and neoplastic disease. Myc proteins are nuclear proteins with relatively short half lives. Amplification of the c-Myc gene has been found in several types of human tumors including lung, breast and colon carcinomas, while the N-Myc gene has been found amplified in neuroblastomas. The L-Myc gene has been reported to be amplified and expressed at high level in human small cell lung carcinomas. The presence of three sequence motifs in the c-Myc COOH terminus, including the leucine zipper, the helix-loop-helix and a basic region provided initial evidence for a sequence-specific binding function. A basic region helix-loop-helix leucine zipper motif (bHLH-Zip) protein, designated Max, specifically associates with c-Myc, N-Myc and L-Myc proteins. The Myc-Max complex binds to DNA in a sequence-specific manner under conditions where neither Max nor Myc exhibit appreciable binding. Max can also form heterodimers with at least two additional bHLH-Zip proteins, Mad and Mxi1, and Mad-Max dimers have been shown to repress transcription through interaction with mSin3. |
Key Feature
| Clonality | Polyclonal |
|---|---|
| Isotype | IgG |
| Host Species | Rabbit |
| Tested Applications | |
recommended for detection of Thr 358 phosphorylated c-Myc of mouse, rat and human origin by WB, IP, IF and IHC(P): |
|
| Species Reactivity | |
| Concentration | 1mg/ml |
| Purification | Affinity purified |
Target Information
| Alternative Names | epitope corresponding to phosphorylated Thr 358 of c-Myc of human origin |
|---|---|
| Tissue Specificity | epitope corresponding to phosphorylated Thr 358 of c-Myc of human origin |
Database Links
| Entrez Gene | 4609 |
|---|
Application
-

Application
Western blot analysis of phosphorylated c-Myc expression in untreated (A) and UV-treated (B) HT-29 whole cell lysates.
-

Application
Immunoperoxidase staining of formalin-fixed, paraffin-embedded human breast carcinoma tissue showing nuclear staining.
| Application Notes | recommended for detection of Thr 358 phosphorylated c-Myc of mouse, rat and human origin by WB, IP, IF and IHC(P): |
|---|
Additional Information
| Form | Liquid |
|---|---|
| Storage Instructions | For short-term storage, store at 4° C. For long-term storage, aliquot and store at -20ºC or below. Avoid multiple freeze-thaw cycles. |
| Storage Buffer | phosphate buffered saline , pH 7.4, 150mM NaCl, 0.02% sodium azide and 50% glycerol. |
Note: The product is for research use only,not for use in diagnostic or therapeutic procedures.
- Related products
- Camel Growth Hormone (GH) ELISA Kit OM642709
- Camel Insulin-like growth factors 1 (IGF-1) ELISA Kit OM642708
- Human HIV-1 p24 core protein (HIV-1 p24) antibody ELISA Kit OM642706
- Antigen Repair 20X pH6.0 OM642703
- 647 labeled Tyramide OM642699
-
- ASSAY KITS
-
- SERUM
Search the catalog number from any major antibody supplier/brand in the search bar above. The search engine will return OmnimAbs products for these targets.
2013 © Omnimabs , All Rights Reserved.

